

Aromatic compounds containing benzene rings are susceptible to attack by electrophiles, that is electron deficient or electron poor species. Common electrophiles include positively charged ions such as: hydrogen ions (H+) and nitronium ions (NO2+) as well as molecules with polar covalent bonds which carry a partial positive charge such as molecules containing the carbonyl group (C=O).
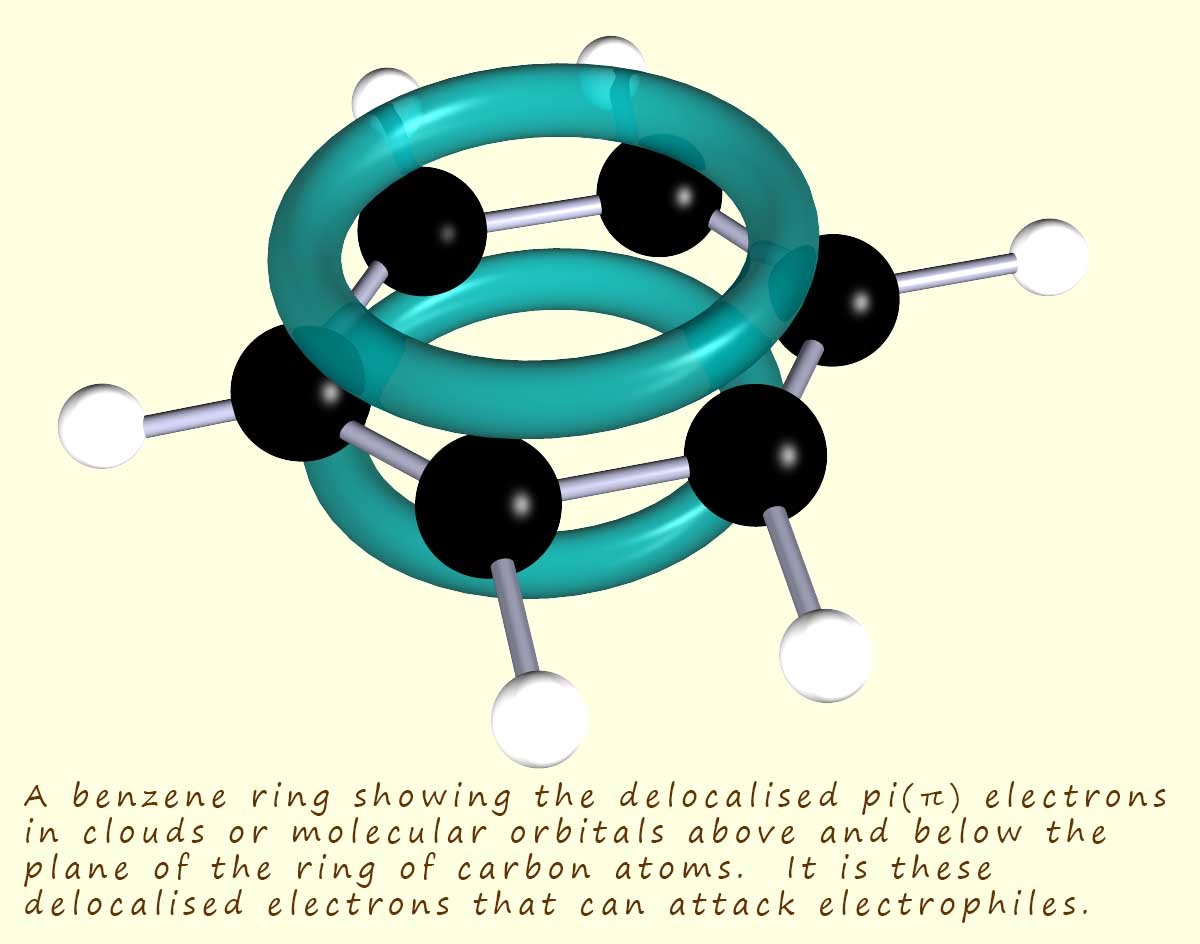
Aromatic compounds containing benzene rings have areas of high electron density or to be more accurate molecular orbitals containing delocalised pi (π) electrons above and below the flat plane of carbon atoms; as shown in the image opposite. These clouds of electron density attract electrophiles, which will act as electron acceptors and readily form bonds with the delocalised pi(π) electrons in the benzene ring.
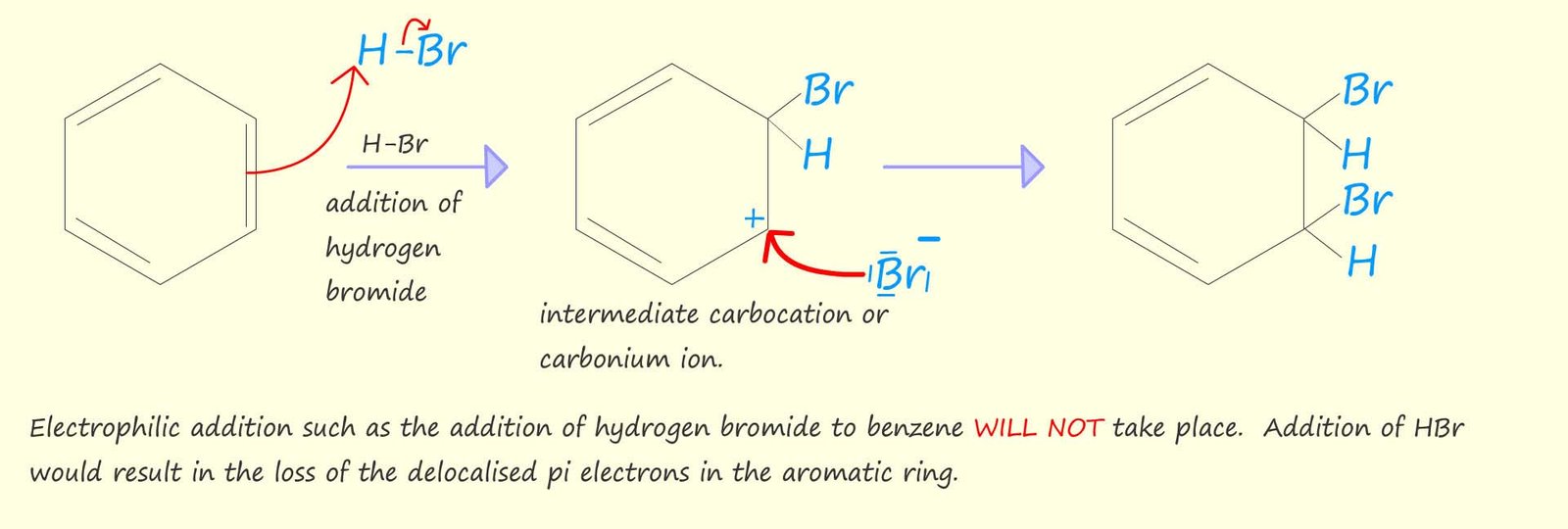
The molecular formula of benzene (C6H6) would suggest that it is an unsaturated molecule and therefore you might expect it to undergo electrophilic addition reactions which are typical of unsaturated molecules such as the alkenes. However you may recall that the presence of a delocalised system of pi (π) electrons within benzene rings adds extra stability and makes benzene and other aromatic molecules much more stable that might be expected.
Simple electrophilic addition to the benzene ring would destroy this system of delocalised
pi (π) electrons and as a result benzene and other aromatic molecules will not undergo electrophilic addition reactions, this is outlined in the image below:
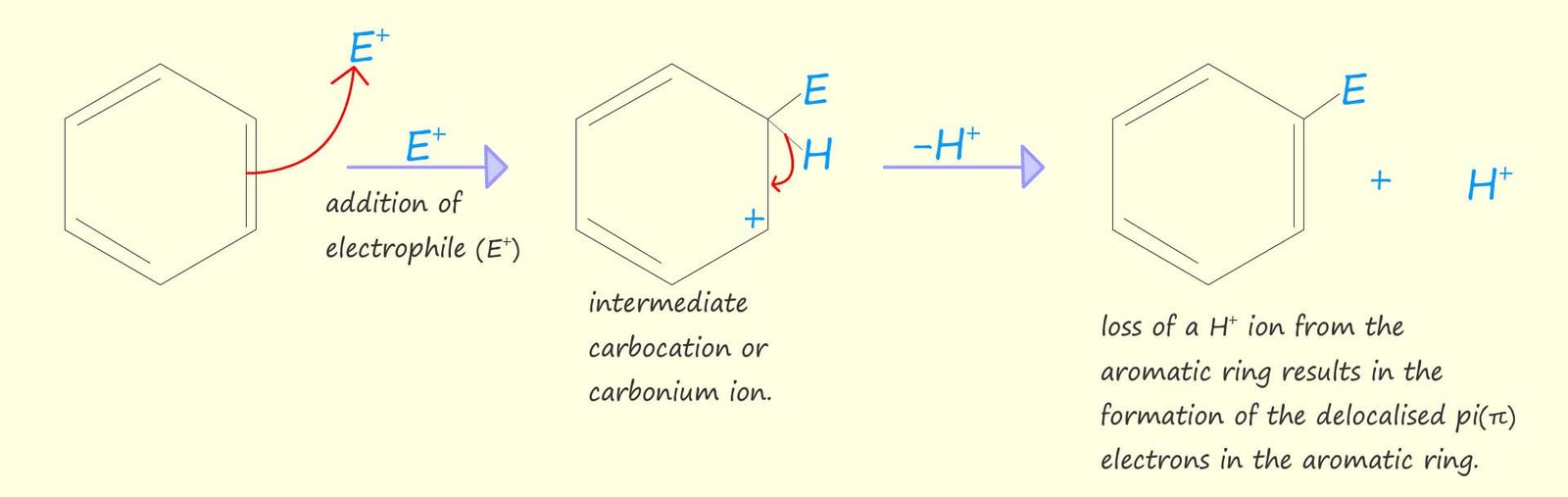
 Instead aromatic molecules such as benzene will undergo electrophilic substitution reactions. The overall result of an electrophilic substitution reaction is that one of the hydrogen atoms present in the aromatic molecule such as benzene is replaced or substituted by the electrophile. Electrophilic substitution reactions unlike electrophilic addition reactions leave the delocalised pi (π) electron system intact. We can show this electrophilic substitution reaction using the equations below:
Instead aromatic molecules such as benzene will undergo electrophilic substitution reactions. The overall result of an electrophilic substitution reaction is that one of the hydrogen atoms present in the aromatic molecule such as benzene is replaced or substituted by the electrophile. Electrophilic substitution reactions unlike electrophilic addition reactions leave the delocalised pi (π) electron system intact. We can show this electrophilic substitution reaction using the equations below:
Or using the circle notation to represent the aromatic ring we have:
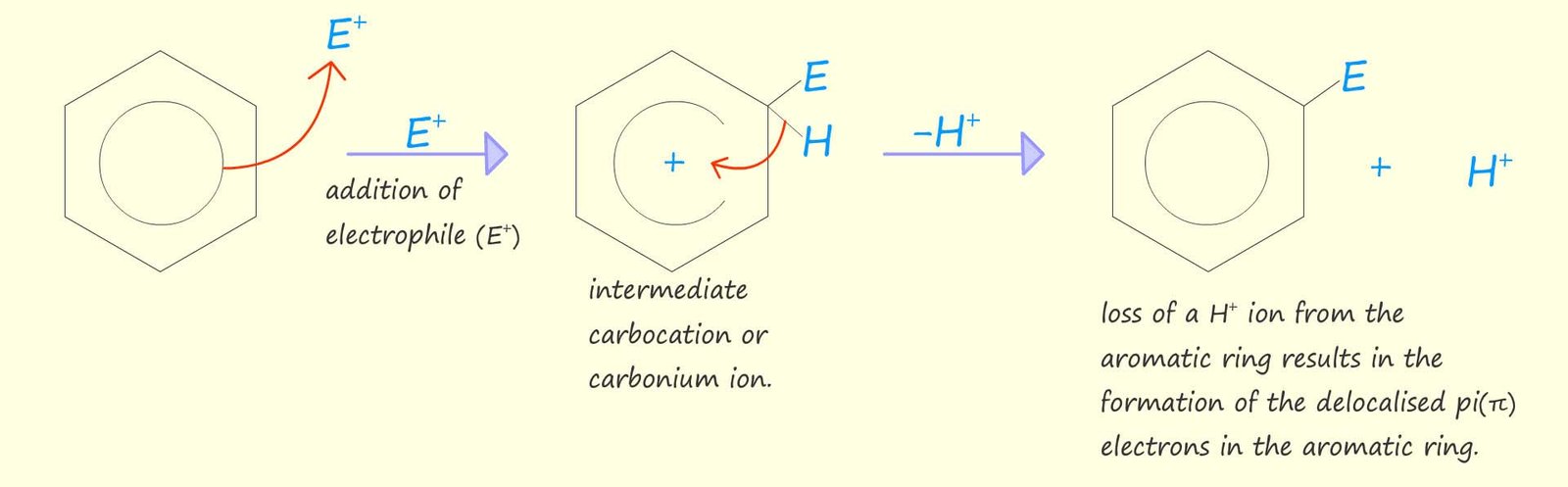
 The mechanism for electrophilic substitution reactions can be thought of as occurring in a number of simple steps:
The mechanism for electrophilic substitution reactions can be thought of as occurring in a number of simple steps:
 The intermediate carbocation formed during an electrophilic substitution reaction will be a resonance stabilised ion.
Addition of the electrophile
to the benzene ring is likely to be the slow step in the above reaction since it will remove
or destroy the aromatic stabilisation
that results from the delocalisation of the pi (π) electrons in the
ring.
The intermediate carbocation formed during an electrophilic substitution reaction will be a resonance stabilised ion.
Addition of the electrophile
to the benzene ring is likely to be the slow step in the above reaction since it will remove
or destroy the aromatic stabilisation
that results from the delocalisation of the pi (π) electrons in the
ring.
Normally when a covalent bond forms the two electrons involved in the bond are held firmly in place between the two nuclei of the atoms forming the covalent bond, however during resonance the electrons involved in forming the covalent bonds are spread out over multiple atoms, the electrons involved in resonance are usually delocalised pi electrons such as those found in aromatic compounds such as benzene.
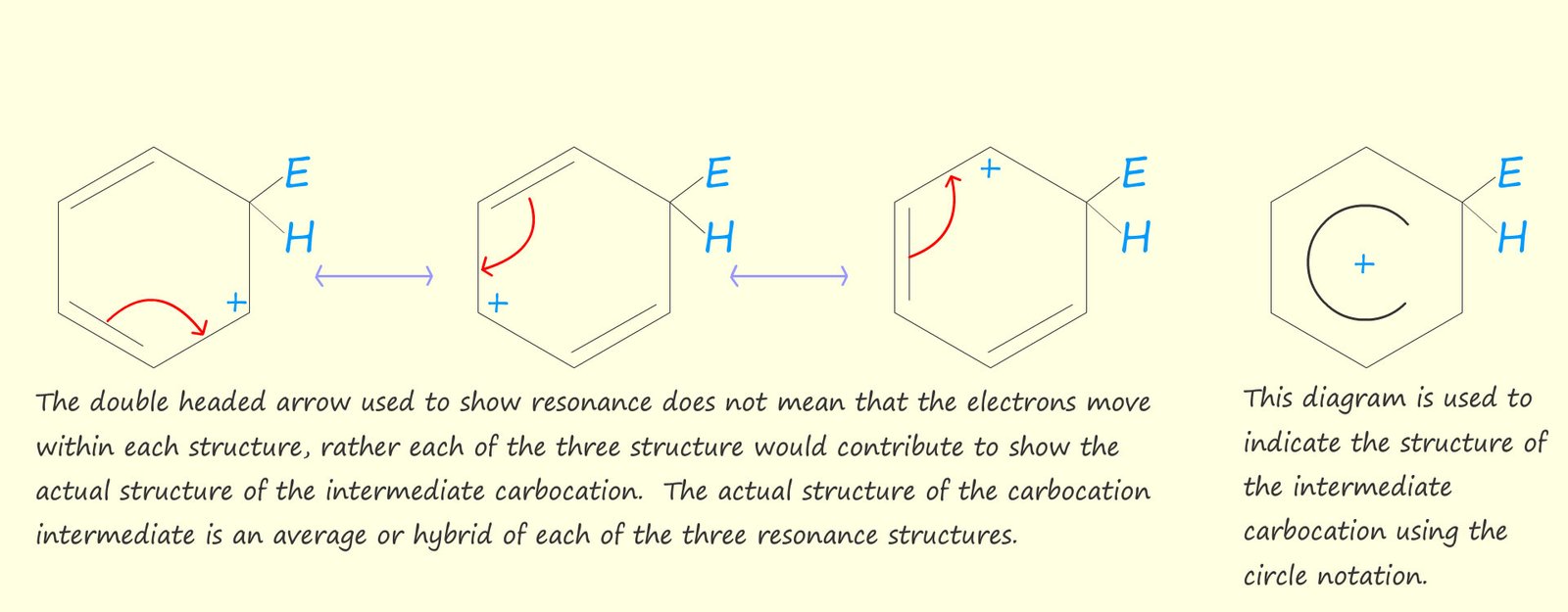
It is not correct to say that delocalised electrons are free to move within a molecule, but rather the delocalised electrons are spread out over multiple atoms. The ability of a molecule or ion to form many different resonance structures will provide additional stability to the molecule or ion. In the diagram below it is possible to draw three resonance structures
for the intermediate carbocation. Generally the more
resonance structures you
are able to draw the more stable the molecule or ion is likely to be.
Electrophilic substitution is a very versatile reaction and it is possible to add a wide variety of substituents onto a benzene ring using this reaction, for example: